Solvent-free approaches to nanoparticles synthesis and polymer functionalization and plasmonic catalytic hydrogenation reactions
Séminaire Chimie IBMM
Dr. Audrey
Le Lundi 20 Mai 2019 à 10h
ENSCM, Amphithéâtre Godechot (campus Balard, 240 av. Émile-Jeanbrau)
Date de début : 2019-05-20 10:00:00
Date de fin : 2019-05-20 11:30:00
Lieu : ENSCM amphi Godechot (campus Balard, 240 av E. Jeanbrau)
Intervenant : Dr. Audrey
Centre for Green Chemistry and Catalysis, McGill University, Montréal, Canada
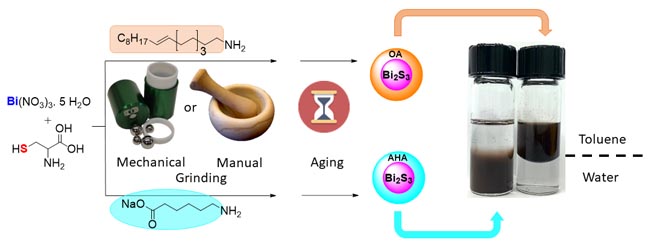
Nanomaterials are intensely researched for their powerful properties applicable in the broad fields of medicine, electronics, optics and catalysis. We developed a novel synthetic method for the scalable production of metal and metal sulfide NPs under solvent-free, mechanochemical conditions. The synthesis of Au NPs provided access to monodisperse and ultra-small NPs in the size range of 1–4 nm, without external reducing agents or bulk solvents.[1] Using lignin as a biomass-based reducer, we could access embedded NPs of Au, Ru, Pd and Re.[2] With Ag, this method gave access to antibacterial filters in a simplified fashion.[3] We also explored the synthesis of metal sulfide with similar Bi2S3 nanoparticles were easily synthesized by mechanochemical activation from molecular precursors and cysteine as a sulfur source, followed by aging to afford X-ray active materials. These materials are exciting candidates for cancer detection.[4] In last examples, I will illustrate how mechanochemistry and aging techniques may be used towards the efficient functional of man-made and biomass based polymers, such as cellulose nanocrystals [5] and chitin.[6]
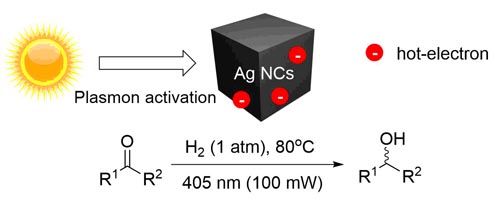
Besides, we have employed silver nanocubes for hydrogen activation and hydrogenation of ketones and aldehydes via irradiation at 405 nm, corresponding to the position of the plasmon band of the nanocubes.[7] Exposure to other wavelengths, or absence of light failed to provide activity thus proving the plasmonic effect. Compared to other catalytic systems, the plasmonically activated catalyst provides access to primary and secondary alcohols using milder conditions, in a highly atom economical fashion.
References
1. M.J. Rak, N.K. Saadé, T. Friščić, A. Moores, Green Chem. 2014, 16, 86–89.
2. M. J. Rak, T. Friščić, A.H. Moores, Faraday Discuss. 2014, 170, 155–167.
3. M.J. Rak, T. Friščić, A. Moores, RSC Adv. 2016, 6, 58365–58370.
4. M.Y. Malca, H. Bao, T. Bastaille, N.K. Saadé, J.M. Kinsella, T. Friščić, A. Moores, Chem. Mater. 2017, 29, 7766–7773.
5. B.G. Fiss, L. Hatherly, R.S. Stein, T. Friščić, A. Moores, ACS Sustainable Chem. Eng. 2019, in press.
6. T. Di Nardo, C. Hadad, A. Nguyen Van Nhien, A. Moores, ChemRxiv 2018, DOI: 10.26434/chemrxiv.7312070.v3
7. M. Landry, A. Gellé, B.Y. Meng, C.J. Barrett, A. Moores, ACS Catal. 2017, 7, 6128–6133.
Contact local IBMM : Dr. Thomas-Xavier