 https://ibmm.umontpellier.fr/wp-content/uploads/Jao_Tanepau.jpg
250
200
Caroline Clavel
https://ibmm2.umontpellier.fr/wp-content/uploads/logo_ibmm_hd.jpg
Caroline Clavel2025-07-11 10:08:082025-07-11 10:08:08Félicitations à Joao pour le prix du meilleur poster !
https://ibmm.umontpellier.fr/wp-content/uploads/Jao_Tanepau.jpg
250
200
Caroline Clavel
https://ibmm2.umontpellier.fr/wp-content/uploads/logo_ibmm_hd.jpg
Caroline Clavel2025-07-11 10:08:082025-07-11 10:08:08Félicitations à Joao pour le prix du meilleur poster !- En savoir +
L’Institut des Biomolécules Max Mousseron est une Unité Mixte de Recherche (UMR 5247) UM, CNRS, ENSCM qui développe des recherches à l’interface de la chimie, de la biologie et de la santé intégrant les enjeux du développement durable : nourrir, soigner, protéger.

Actualités
 https://ibmm.umontpellier.fr/wp-content/uploads/Jao_Tanepau.jpg
250
200
Caroline Clavel
https://ibmm2.umontpellier.fr/wp-content/uploads/logo_ibmm_hd.jpg
Caroline Clavel2025-07-11 10:08:082025-07-11 10:08:08Félicitations à Joao pour le prix du meilleur poster !
https://ibmm.umontpellier.fr/wp-content/uploads/Jao_Tanepau.jpg
250
200
Caroline Clavel
https://ibmm2.umontpellier.fr/wp-content/uploads/logo_ibmm_hd.jpg
Caroline Clavel2025-07-11 10:08:082025-07-11 10:08:08Félicitations à Joao pour le prix du meilleur poster !
Morgan Pellerano présente son métier sur le site de l’UM
Morgan a été interviewé pour présenter ses activités, en…
16 équipes de recherche
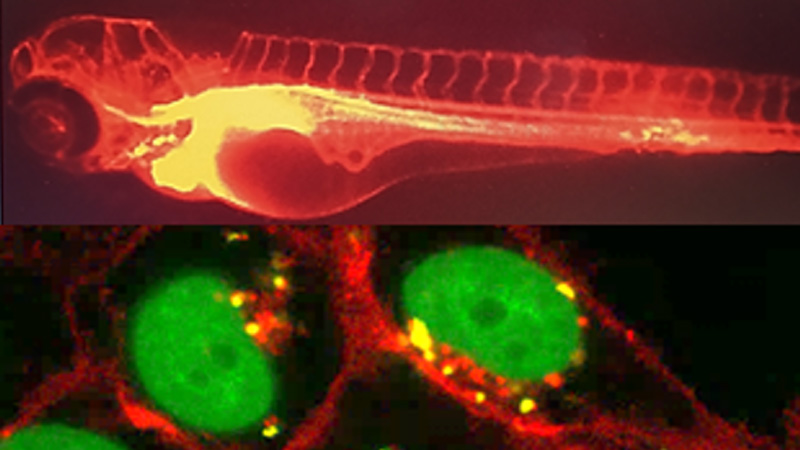
Glyco et Nanovecteurs pour le ciblage Thérapeutique
10 janvier 2024/par Stephanie HERNANDEZ
Chimie Verte et Technologies Innovantes
10 janvier 2024/par Stephanie HERNANDEZ
Synthèse stéréosélective & acides aminés modifiés
10 janvier 2024/par Stephanie HERNANDEZ
Dynamique des Systèmes Biomoléculaires Complexes
10 janvier 2024/par Stephanie HERNANDEZ
Glycochimie & reconnaissance moléculaire
10 janvier 2024/par Stephanie HERNANDEZ
Sciences analytiques des biomolécules
10 janvier 2024/par Stephanie HERNANDEZ
Oncopharmacochimie & Pharmacotoxicologie Cutanée
10 janvier 2024/par Stephanie HERNANDEZ
Supramolecular MAchines and ARchitecture Team
10 janvier 2024/par Stephanie HERNANDEZ
ChemBioNAC
10 janvier 2024/par Stephanie HERNANDEZ
Acides aminés, hétérocycles, peptides & protéines
10 janvier 2024/par Stephanie HERNANDEZ
Pharmacologie Cellulaire
10 janvier 2024/par Stephanie HERNANDEZ
Pharmacochimie, transmission synaptique & neuroprotection
10 janvier 2024/par Stephanie HERNANDEZ
Oncothérapie et oncopharmacologie
10 janvier 2024/par Stephanie HERNANDEZ
Nucléosides & Effecteurs Phosphorylés
10 janvier 2024/par Stephanie HERNANDEZ
Synthèse de Lipides Bioactifs
10 janvier 2024/par Stephanie HERNANDEZ







Offre de thèse : Revisiting the Amidation Reaction by Flow Mechanochemistry and Machine Learning
Context and objectives: Mechanochemistry and flow chemistry have been recognized in 2019 by the International Union for Pure and Applied Chemistry (IUPAC) as two of the emerging technologies that could contribute to the sustainable development of our planet by reducing the carbon footprint.1 By merging both areas, reactive extrusion known also as flow mechanochemistry provides […]
Offre de thèse : PHOTOCLICK: Development of biorthogonal photo-crosslinking reactions for 3D printing micro-structured biomaterials
Contract: 3 years starting from October 2025 Application deadline: 04/07/2025 Location: Institute of Biomolecules Max Mousseron (IBMM), Montpellier, France and Institute for Molecular Systems Engineering and Advanced Materials (IMSEAM), Heidelberg, Germany The project This thesis project is located at the interface of click chemistry, photochemistry, materials, 3D printing and biology. The goal is to […]